9C.2.1.2 Chemical Technology & Organic Compounds
Explain the political, societal, economic and environmental impact of chemical products and technologies.
For example: Pollution effects, atmospheric changes, petroleum products, material use or waste disposal.
Compare and contrast the structure, properties and uses of organic compounds, such as hydrocarbons, alcohols, sugars, fats and proteins.
Overview
MN Standard in Lay Terms
Organic chemistry is the chemistry of carbon compounds formed by covalent bonding. The reactions and properties of the carbon compound are largely determined by the types of atoms and groups of atoms (functional groups) bonded to the carbon.
The development of synthetic organic compounds, particularly from fossil fuels, has greatly impacted modern society providing both positive benefits and challenges.
Big Idea
Because of its four valence electrons, carbon has an ability to bond with itself and other atoms by sharing electrons in a variety of ways including single, double, and triple bonds. Carbon can also bond to groups of atoms (a functional group) which determine the properties and reactions of the molecules formed.
The ability of chemists to synthetically produce organic compounds has spawned a modern consumer products industry with its associated environmental and societal challenges.
MN Standard Benchmarks
9C.1.3.3.1 Explain the political, societal, economic and environmental impact of chemical products and technologies.
9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins.
The Essentials

Cartoon from this site.
Structure and properties of matter
12BPS2.6 Carbon atoms can bond to one another in chains, rings, and branching networks to form a variety of structures, including synthetic polymers, oils, and the large molecules essential to life. (See above suggestions for ideas on tying current research ideas into relevant green chemistry lessons)
Natural resources
12FSPSP3.1 Human populations use resources in the environment in order to maintain and improve their existence. Natural resources have been and will continue to be used to maintain human populations.
12FSPSP3.2 The earth does not have infinite resources; increasing human consumption places severe stress on the natural processes that renew some resources, and it depletes those resources that cannot be renewed.
12FSPSP3.3 Humans use many natural systems as resources. Natural systems have the capacity to reuse waste, but that capacity is limited. Natural systems can change to an extent that exceeds the limits of organisms to adapt naturally or humans to adapt technologically.
The interdependence of organisms
12CLS4.4 Living organisms have the capacity to produce populations of infinite size, but environments and resources are finite. This fundamental tension has profound effects on the interactions between organisms.
12CLS4.5 Human beings live within the world's ecosystems. Increasingly, humans modify ecosystems as a result of population growth, technology, and consumption. Human destruction of habitats through direct harvesting, pollution, atmospheric changes, and other factors is threatening current global stability, and if not addressed, ecosystems will be irreversibly affected.
Physical Setting - Chemical Reactions
The configuration of atoms in a molecule determines the molecule's properties. Shapes are particularly important in how large molecules interact with others. 4D/H8
Benchmarks of Science Literacy
The configuration of atoms in a molecule determines the molecule's properties. Shapes are particularly important in how large molecules interact with others. 4D/H8
Sunlight is the ultimate source of most of the energy we use. The energy in fossil fuels such as oil and coal comes from energy that plants captured from the sun long ago. 8C/H8** (BSL)
NAEP
P12.7: A large number of important reactions involve the transfer of either electrons (oxidation/reduction reactions) or hydrogen ions (acid/base reactions) between reacting ions, molecules, or atoms. In other chemical reactions, atoms interact with one another by sharing electrons to create a bond. An important example is carbon atoms, which can bond to one another in chains, rings, and branching networks to form, along with other kinds of atoms (hydrogen, oxygen, nitrogen, and sulfur), a variety of structures, including synthetic polymers, oils, and the large molecules essential to life.
Common Core Standards
2010 Literacy Standards - Reading Benchmarks: Literacy in Science and Technical Subjects 6-12
Integration of Knowledge and Ideas (Benchmark 11.13.7.7) Integrate and evaluate multiple sources of information presented in diverse formats and media (e.g., quantitative data, video, multimedia) in order to address a question or solve a problem.
Common Core Language Arts Standards: Students can write a laboratory report in the proper form and using their knowledge of technical writing skills. Common Core Standards addressed:
RST.9-10-1. Cite specific textual evidence to support analysis of science and technical texts, attending to the precise details of explanations or directions.
RST.9-10-2. Determine the central ideas or conclusions of a text; trace the text's explanation or description of a complex process, phenomena or concept; provide an accurate summary of the text.
RST.9-10.3. Follow precisely a complex multistep procedure when carrying out experiments; taking measurements or performing technical tasks, attending to special cases or exceptions defined in the texts.
Misconceptions
1. Students fail to recognize the three dimensional nature of carbon compounds. For example, in naming a branched alkane they don't realize that the carbon atoms in the straight chain may be counted from either end of the molecule. Students think that a "branch" is on a different numbered carbon but the molecule can be "flipped over" and counted from the other end. This is why the carbon atoms are counted from either end that gives the branch the shortest number.
Vignette
Ms. Apple begins her unit on the "chemistry of carbon" by dividing students into groups of four and giving each group a "puzzle". She hands each group a baggie with six "carbon atoms" and 14 "hydrogen atoms." The atoms are circles of paper with the symbol of the element and lines indicating the number of bonds each can form. For example carbon would be
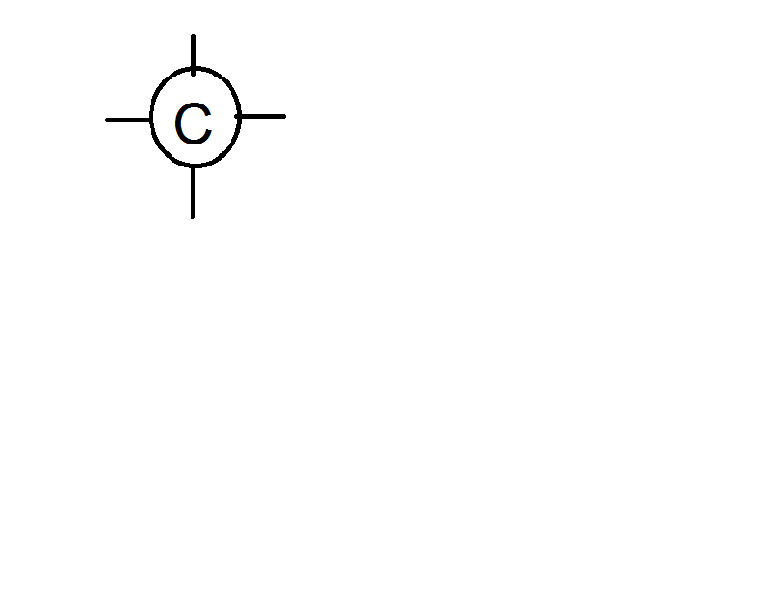 and hydrogen would have a circle with only one line. Students are to arrange the atoms into molecules and then write the "structural" formula of the molecule by writing the number of hydrogen atoms on each carbon and showing the arrangements of the carbon atoms. For example an arrangement of all of the carbon atoms in a row would be CH3-CH2-CH2-CH2-CH2-CH3. Another possible arrangement could be:
and hydrogen would have a circle with only one line. Students are to arrange the atoms into molecules and then write the "structural" formula of the molecule by writing the number of hydrogen atoms on each carbon and showing the arrangements of the carbon atoms. For example an arrangement of all of the carbon atoms in a row would be CH3-CH2-CH2-CH2-CH2-CH3. Another possible arrangement could be: 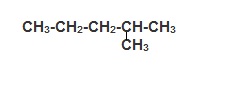 Students continue working and recording their possible structures. Ms. Apple moves among the students and guides them with questions about whether two structures are indeed different.
Students continue working and recording their possible structures. Ms. Apple moves among the students and guides them with questions about whether two structures are indeed different.
When students have had time to draw several molecules Ms. Apple asks the students to give her the chemical formula for each molecule. Students then realize that every molecule has the formula C6H14. She then explains how structural formulas and molecular formulas differ. She also introduces the term "isomers." Students are asked to examine their structures to determine which molecules are true isomers and which might actually be arrangements of atoms that give same structure. The class determines there are only five different isomers of hexane.
Ms. Apple also provides students with the formulas of other similar hydrocarbons. She has them examine the number of carbon and hydrogen atoms in each to see if there is a pattern. Students arrive at the general formula for a hydrocarbon with single bonds called "alkanes" (CnH2n+2). She has her students view a video with an excellent overview of "organic" chemistry World of Chemistry Video 21: Chemistry of Carbon Ms. Apple also has students research the properties and uses of hydrocarbons as fuels.
Later Ms. Apple introduces a project she calls "How Gasoline Drives Our World." Students are placed into groups of 3-4. Each group is to pose a question about gasoline related to its production, use, economics, environmental impact or role in world politics. Groups then research their answer and collect background information. Groups present their information with a short oral report to the class. A Closer Look At Energy and Oil Consumption Ms. Apple has found this project to be an excellent method for students to engage in the Nature of Science and Engineering standard (Benchmark: 9C.1.3.3 Explain the political, societal, economic and environmental impact of chemical products and technologies).
Note: Here is an "alternative" project for this Nature of Science and Engineering benchmark. Minnesota has a huge industry based on alcohol fermentation and potential "biofuels." Students research the potential economic, environmental and political "barriers" to these fuels and present their information in an oral report to the class.
In the second half of this unit Ms. Apple gives each group a molecular model kit and then asks students to construct molecules of other organic compounds. (Note: students who have already taken Biology have worked on these topics in Benchmark 9.4.1.2.1- Recognize that cells are composed primarily of a few elements (carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur), and describe the basic molecular structures and the primary functions of carbohydrates, lipids, proteins and nucleic acids.)
Students are given a handout which reviews the formula of functional groups such as alcohols and composition of organic compounds such as sugars, fats, and proteins. Students build a molecular model of each type of organic compound. They research the properties of each organic compound and their interactions in reactions such as dehydration and synthesis. (Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins.)
Resources
Instructional suggestions/options
Molecular model kits are an excellent tool for assisting students in visualizing the structures of organic compounds. Students will better understand the arrangement of atoms in an organic compound if they "build" the compound using a molecular model kit.
Current research deals a lot with making chemistry "greener" particularly using plants and other bio-waste to make fuels. Chemmatters has an article, Green Gasoline, Fuel from Plants is readable for all levels and relevant.
Modern research in biochemistry (health and medicines, fuel sources, plastics, etc) is easily tapped into here to add to the current dilemma scientists face as they work with environmental groups.
Suggested Labs and Activities
This activity tests for carbohydrates, proteins and lipids. Students look at how food is chemistry.
Polymers - Make and study a polymer in order to understand it's properties.
Carbohydrates Lab Activity Identifying Carbohydrates
(Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins. This activity is a lab by which students use chemical methods to identify types of sugars and starch).
Identifying types of Sugars Lab Activity Lab: Carbohydrates Testing with Chemical Indicators
(Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins ). This activity is a lab by which students use chemical methods to identify types of sugars and starch.
Science NetLinks: the Chemistry of Petroleum and Hydrocarbons What are Hydrocarbons Lessons and Labs
(Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins). This website provides lesson plans on the basics of hydrocarbons. It includes information for students and an excellent video and interactive simulation of crude oil refining.
Lesson Plans on Organic Chemistry Lesson Planet- Search Engine for Science Lesson Plans This is a commercial website which provides organic chemistry labs.
(Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins.)
Free Lessons and Lab Activities on Hydrocarbons and Organic Chemistry ChalkBored
(Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins). This site provides freeware which allows the user to draw the structures of organic compounds.
Organic Chemistry Lessons Chemistry Zone
(Benchmark: 9C.2.1.2.2 Compare and contrast the structure, properties and uses of organic compounds such as hydrocarbons, alcohols, sugars, fats and proteins.) This site provides tutorials on topics within organic chemistry such as isomers of hydrocarbons, polymers and nomenclature "flashcards".
Additional resources
Uncovering Student Ideas in Science
Volume 4: Where does Oil Come From - pages 151-156
World of Chemistry videos # 21 Chemistry of Carbon, # 22 The Age of Polymers and # 23 Proteins: Structure and Function World of Chemistry Video Series
Molecular Model KitsFlinn Molecular Model Kits
Flinn Lab Manual For Organic Chemistry Flinn Chemtopics Lab Manual # 19
Chemistry In The Community (Chem Com) has excellent petroleum and food unit. ChemCom resources
Discovery Elements Video and resources Elements of Chemistry - Carbon Chemistry VHS
Science Literacy:
The April 2011 issue of ChemMatters celebrates the second theme of the International Year of Chemistry 2011: "Alternative Sources of Energy," with articles on how hydrogen may one day replace gasoline in cars and how old cooking oil can be used as a cheap and more environmentally friendly substitute to gasoline in cars.
Chemmatters - Articles by Topic is a good starting point for current, readable information for general chemistry courses
Vocabulary/Glossary:
- Hydrocarbon :A compound containing only the elements hydrogen and carbon. The carbon atoms may have single, double or triple bonds between them.
- Alcohol: An organic compound containing one or more hydroxyl ( -OH) groups on the molecule.
- Sugar: A subgroup of carbohydrates consisting of one or more unit of C6H12O6.
- Fats: A class of biochemical compounds containing an acid group (COOH) on one end of the molecule and a long hydrocarbon chain on the other end. Fats which do not have carbons with multiple bonds are said to be saturated.
- Amino Acid - A compound containing an organic acid functional group (COOH) at one end and amine functional group (NH2) at the other end.
- Proteins: An organic biological polymer that is made up of polypeptide chains of 50 or more amino acids.
- Isomers: Each of two or more compounds with the same formula but a different arrangement of atoms in the molecule and different properties.
- Functional Group: A group of atoms responsible for the characteristic behavior of the class of compounds in which the group occurs, as the hydroxyl group (OH) in alcohols.
Minnesota 9-12 Life Science Standard: Benchmark 9.4.1.2.1Recognize that cells are composed primarily of a few elements (carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur), and describe the basic molecular structures and the primary functions of carbohydrates, lipids, proteins and nucleic acids.
Organic chemistry has connections with Social Studies with the historical development and use of fossil fuels during the Industrial Revolution and Twentieth Century.
Assessment
Assessment of Students
What molecule is removed when a glucose and fructose molecule are linked together to form a sucrose (table sugar)?. (taxonomic level: application).
Why can we get more energy from complex hydrocarbons than alcohols?
Some organic compounds burn hotter and cleaner than others. Why?
Draw the condensed structure of the two isomers of butane (C4H10). (taxonomic level: application)
Explain how carbon's bonding ability allows it to become the backbone of an entire discipline of chemistry called "organic" chemistry? (taxonomic level: application)
Explain the difference between the two types of sugars like glucose and sucrose. (taxonomic level analysis)
What is the difference in the bonding between carbon atoms in "saturated fats" as opposed to "unsaturated fats"? (taxonomic level: knowledge)
Assessment of Teachers
How do each of the key functional groups (alcohols, amino acids, sugars and fatty acids/glycerol)? differ and determine the chemical and physical properties of their molecules?
How might you help students understand the "three dimensional" nature of organic molecules especially when identifying whether two structures of a molecule are different isomers?
What are some consumer products and applications of organic chemistry from your own everyday experiences?
Differentiation
Struggling and At-Risk
"At risk" students will learn more if they believe the content is "relevant" to their lives.
These lab activities will really help students learn about polymers. They are great for in the classroom and some of them can even be done at home with the help of a parent. So see what it's like to do some science with polymers! Polymer Fun
From Teaching Science To ELL's NSTA Journal The Science Teacher March 2011
Provide opportunities for interaction for ELLs' an activity that connects input and output. Interaction serves as the foundation for language development because it requires ELLs to comprehend language input, produce language output, and negotiate meaning with others. Through interaction, ELLs not only develop their scientific communication skills, but also clarify their understanding with classmates who are more proficient in English. Teaching Strategies for ELLs by Nazan Bautista and Marcha Castaneda The Science Teacher March 2011.
Have students research the difference between "saturated" and "unsaturated" hydrocarbons and how these terms apply to fats as well. Students can give a short oral report to the class explaining the differences and similarities of saturated and unsaturated fats and their impact on personal health.
Have students research how Haiti an island nation has no known source of fossil fuels. As a result the Haitian people have had to resort to using charcoal from tree wood for cooking. This has caused large deforestation of the rain forest in Haiti.
Parents/Admin
Administrators
Administrators should observe students engaged in using molecular model kits to build molecules while comparing and contrasting the structures of various organic compounds.
Because organic chemistry is an important component of many consumer products, administrators would also observe the teacher assisting students in making the connections between organic chemistry and their daily experiences.
Parents can support their student's understanding of the applications of organic chemistry pointing out household examples of organic compounds. These might include hydrocarbon fuels, consumer products containing alcohols and food products including sugars and fats. Encourage your student to read the labels of household products.
