6.2.3.2 Transformation
Differentiate between kinetic and potential energy and analyze situations where kinetic energy is converted to potential energy and vice versa.
Trace the changes of energy forms, including thermal, electrical, chemical, mechanical or others as energy is used in devices.
For example: A bicycle, light bulb or automobile.
Describe how heat energy is transferred in conduction, convection and radiation.
Overview
Minnesota Standard in Lay Terms
Energy can take many forms: light, electrical, thermal, gravitational, elastic, and chemical energy. Whenever energy appears in one place, it must have disappeared from another. Whenever energy is lost from somewhere, it must have gone somewhere else. Whenever energy is changed from one form to another form (electrical to light, for example) some if it is likely to go into heat and is not available for use. (paraphrased from Atlas of Scientific Literacy)
Big Idea
Forms of energy: Energy can take the form of light, electrical, thermal, gravitational, elastic, and chemical energy. Objects and substances in motion have kinetic energy. Three forms of potential energy are gravitational, elastic, and chemical. Gravitational potential energy changes in a system as the relative positions of objects are changed. Objects can have elastic potential energy due to their compression, or chemical potential energy due to the nature and arrangement of atoms. (NAEP, 2009 p.8.9.)
Conservation: Energy is not lost during transfer or transformation. "Whenever energy appears in one place, it must have disappeared from another. Whenever energy is lost from somewhere, it must have gone somewhere else. Sometime when energy appears to be lost, it actually has been transferred to a system that is so large that the effect of the transferred energy is imperceptible." (Atlas of Scientific Literacy, p. 25, 4E/M1)
Dissipation: "Whenever there is a transformation of energy, some if it is likely to go into heat, which spreads around and is therefore not available for use." (Benchmarks for Science Literacy. p.81).
6.2.3.2.1: Differentiate between kinetic and potential energy and analyze situations where kinetic energy is converted to potential energy and vice versa.
6.2.3.2.2: Trace the changes of energy forms, including thermal, electrical, chemical, mechanical, or others as energy is used in devices.
6.2.3.2.3: Describe how heat energy is transferred in conduction, convection, and radiation.
Correlations
- NSES Standards: Content Standard B: As a result of their activities in grades 5-8, all students should develop an understanding of properties and changes of properties in matter, motions, and forces, transfer of energy. NSES, p.155.
- Atlas of Science Literacy: pp. 24-25
- Benchmarks of Science Literacy 4E, 6-8.
By the end of 8th grade, students should know that:
- Energy cannot be created or destroyed, but only changed from one form into another
- Most of what goes on in the universe-from exploding stars and biological growth to the operation of machines and the motion of people-involves some form of energy being transformed into another. Energy in the form of heat is almost always one of the products of an energy transformation.
- Heat can be transferred through materials by the collision of atoms or across space by radiation. If the material is fluid, currents will be set up in it that aid the transfer of heat.
- Energy appears in different forms. Heat energy is in the disorderly motion of molecules; chemical energy is in the arrangement of atoms; mechanical energy is in moving bodies or in elastically distorted shapes; gravitational energy is in the separation of mutually attracting masses.
Common Core Standards
- Language Arts: Comprehension and Collaboration
- Engage effectively in a range of collaborative discussions (one-on-one, in groups, and teacher-led) with diverse partners on grade 6 topics, texts, and issues, building on others' ideas and expressing their own clearly.
- Math: Develop understanding of statistical variability
- Summarize numerical data sets in relation to their context, such as describing the nature of the attribute under investigation, including how it was measured and its units of measurement.
Misconceptions
Student Misconceptions
- [Often] cold is thought to be transferred rather than heat.
- Some materials may be thought to be intrinsically warm (blankets) or cold (metals).
- Objects that keep things warm, such as a sweater or mittens, may be thought to be sources of heat. (Benchmarks for Scientific Literacy, p. 83)
- Some students think that "cold" is being transferred from a colder to a warmer object; others that both "heat" and "cold" are transferred at the same time.
- Students often think objects cool down or release heat spontaneously-that is, without being in contact with a cooler object (Kesidou, 1990; Wiser, 1986). Even after instruction, students don't always give up their naive notion that some substances (for example, flour, sugar, or air) cannot heat up (Tiberghien, 1985) or that metals get hot quickly because "they attract heat," "suck heat in," or "hold heat well" (Erickson, 1985).
- Middle-school students believe different materials in the same surroundings have different temperatures if they feel different (for example, metal feels colder than wood). [Students] do not recognize the universal tendency to temperature equalization (Tomasini and Balandi, 1987).
- Few middle- and high-school students understand the molecular basis of heat transfer even after instruction (Wiser, 1986; Kesidou and Duit, 1993). Although specially designed instruction appears to give students a better understanding about heat transfer than traditional instruction, some difficulties often remain (Tiberghien, 1985; Lewis, 1991)." (Benchmarks for Scientific Literacy, pp. 337-338)
- Middle- and high-school students tend to think that energy transformations involve only one form of energy at a time (Brook and Wells, 1988).
- Although [students] develop some skill in identifying different forms of energy, in most cases their descriptions of energy change focus only on forms that have perceivable effects (Brook and Driver, 1986).
- The transformation of motion to heat seems to be difficult for students to accept, especially in cases with no obvious temperature increase (Brook and Driver, 1986; Kesidou & Duit, 1993).
- Finally, it may not be clear to students that some forms of energy, such as light, sound, and chemical energy, can be used to make things happen (Carr & Kirkwood, 1988)." Benchmarks of Science Literacy, p. 338.
Vignette
(6.2.3.2.2) In the Engineering Is Elementary (EIE) Kit Electrical Engineering: Designing Alarm Circuits, students go on a scavenger hunt to find all the ways they use electricity in one day. Each student organizes the data on a T-chart with the technology that uses electricity on the left side and what it does on the right. For example, an alarm clock on the left, to wake someone up on the right.
The class shares their findings together, reporting about 10-12 items from various students and how they are used, with the teacher recording the items on index cards and posting them on the board. The teacher encourages the students to think about non-obvious ways that they use electricity (rather than just TV and radio) and closes this section by guiding the discussion to how electricity impacts their daily lives.
As a class, the 10-12 items that were shared and recorded on index cards are now sorted. Through careful questioning, the students divide the items into 4 groups: 1) things that light, 2) things that get hot, 3) things that make noise, 4) things that move. The teacher puts these 4 categories on 4 pieces of chart paper and posts them around the room. Each student is given 5 post-its and directed to put a technology on each that hasn't been listed yet and post on the appropriate paper. At their tables, the students sort their technologies into the proper categories. When someone discovers that an item should be in more than one, more cards are distributed so that it can be put into each category needed.
As a class, the tables report on one or two items and where they placed them. The teacher asks the class to think about how they would explain electricity to a friend or relative. Their own definitions are compared to a previous one from earlier in the unit. A discussion of the word "energy" emerges and the teacher explains to the students that electricity is only one form of energy that exists. He then goes to the chart paper topics and changes the titles to Light energy, Heat energy, Sound energy, and Mechanical (motion) energy. The teacher goes on to explain that the technologies they have put in those categories transforms the electrical energy into another form.
On chart paper at each table, the teacher has the students create concept maps in groups with their index cards. A bubble with electrical energy in it is in the center with the four other energy forms around the outside. The students place their cards on the appropriate arrows to show which transformation takes place.
Resources
Suggested Labs and Activities
- Benchmark 6.2.3.2.1:
A NASA Video Clip about calculating the Potential and Kinetic Energy of an object. Use the video as a way to explain or elaborate on the concepts of potential or kinetic energy. Teachers might show the video twice. The first time, students are watching for examples of the two types of energy. In preparation for the second viewing, students can make a "T-Chart" in their science notebooks. Students should label one side "kinetic energy" and the other side "potential energy." Watch the video again and record examples of each energy type.
- Benchmark 6.2.3.2.2
Trace the changes of energy forms, including thermal, electrical, chemical, mechanical or others as energy is used in devices. Use this activity to reinforce the concept of energy transformations. Transfer of potential to kinetic energy lab (multiple day activity)
- Benchmark 6.2.3.2.3; Introductory activity:
Describe how heat energy is transferred in conduction, convection, and radiation. Use this activity to introduce the concept of heat transfer. Transfer of heat energy lab
- Benchmark 6.2.3.2.3; Extension activity:
Describe how heat energy is transferred in conduction, convection, and radiation. Thermal conductivity lab
- Benchmark 6.2.3.2.3; Extension activity:
Describe how heat energy is transferred in conduction, convection, and radiation. Use this activity for students needing practice with the concept. Heat Transfer Lab
- Convection: A Current Event, GEMS:
Students explore this important physical phenomenon by observing and charting the convection currents in a liquid.
- Benchmark 6.2.3.2.3; Demonstration activities:
Describe how heat energy is transferred in conduction, convection, and radiation. Use these activities to introduce the benchmark:
Instructional Suggestions/Options
- From: Massachusetts Science and Technology/Engineering Curriculum Framework, October 2006:
In grades 6-8, students still need concrete, physical-world experiences to help them develop concepts associated with motion, mass, volume, and energy. As they learn to make accurate measurements using a variety of instruments, their experiments become more quantitative and their physical models more precise. Students in these grades are able to graph one measurement in relation to another, such as temperature change over time. They may collect data by using microcomputer- or calculator- based laboratories (MBL or CBL), and can learn to make sense immediately of graphical and other abstract representations essential to scientific understanding.
- From: Driver, Rosalind, Squires, Ann, Rushworth, Peter, & Wood-Robinson, Valerie (1994) Making Sense of Secondary Science New York: Routledge. p.147:
Duit suggests that in order to promote learning about energy 'more time should be devoted to qualitative questions,' and that 'students should be advised to explain the physical phenomena in their own words.'
Vocabulary/Glossary
- Conduction: the process by which heat or electricity is directly transmitted through a substance without movement of the material.
- Conservation: the principle by which the total value of a physical quantity (such as energy or mass) remains constant in a system.
- Convection: the movement caused by the tendency of hotter and less dense material to rise, and colder, denser material to sink under the influence of gravity.
- Energy: power derived from the utilization of physical or chemical resources, esp. to provide light and heat or to work machines.
- Force: the push or pull on an object.
- Joule: the SI unit of work or energy.
- Mechanical: energy that is possessed by an object due to its motion or due to its position.
- Radiation: the emission of energy as electromagnetic waves.
- Source: process by which energy or a particular component enters a system.
- System: a set of things working together as parts of a mechanism or as an interconnecting network.
- Thermal: relating to heat.
- Transfer: move (someone or something) from one place to another.
6.2.3.2.1 and 6.2.3.2.2:
Learn about conservation of energy with a skate boarder. Build tracks, ramps, and jumps for the skater and view the kinetic energy, potential energy, and friction as he moves. You can also take the skater to different planets, even outer space!
- Math: Calculate the amount of potential energy of an object at varying heights. Drop the object from predetermined heights and calculate the amount of kinetic energy. Compare the two numbers. Are they exact? Why or why not?
- Social Studies: Study forms of alternative energy and research places on the globe that are actively exploring or experimenting with these energy sources. Determine why different energy sources are popular in some areas but not in others.
- Language Arts: Write and record a podcast that encourages citizens in a town to utilize alternative energy resources. Explain how alternative energy resources would impact life in the town.
Additional Resources
Introduction to Energy (Student Article)
Energy In Depth (website for students and teachers)
Assessment
Assessment of Students
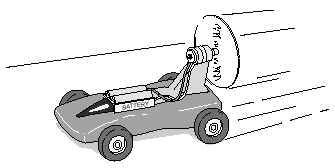
- Summative:
- What energy transformations and forces influence this battery-powered toy car pictured above? Explain your answer fully. (Knowledge, application)
- Formative:
- "Rolling to a Stop." Keeley, P., & Harrington, R. (2010). Uncovering student ideas in physical science: 45 new forces and motion assessment probes. Arlington, VA: National Science Teachers Association. p. 91. (Application)
- Describe the energy transfer in a bouncing ball. (Evaluation)
- Windmills are used to convert wind energy into a more useful form. In most cases, there are three steps in this process. The energy is in a different form at each step. Draw a flow chart that accurately identifies the three steps, in their appropriate order. (Knowledge, synthesis, application, evaluation)
Assessment of Teachers
- How can children's misconceptions about energy act as a stepping-stone to greater scientific understanding?
- Why is it important for students to identify energy transformations?
- How do you determine whether or not a student clearly understands energy transformation?
Differentiation
Struggling and At-Risk
This website provides students an opportunity to design their own roller coaster. Provide students time to "play" on the site and encourage them to create a roller coaster that works. (The cart must stay on the track and move forward until the end of the "ride" in order to be considered a working coaster.) After the coaster has been created, students should sketch it in their science journals. Offer time for students to compare their sketches with their classmates and look for similarities. Provide time for students to label the potential and kinetic energy and promote a discussion about the transfer of energies on the ride.
- "Design a mobile for at least six different energy transfers, including every day examples. Illustrate each type." Westphal, L.E. (2009). Differentiating instruction with menus: science. Waco, Texas: Prufrock Press, Inc.
- While developing Energy concepts with ELL students make sure to illustrate and diagram concepts with vocabulary on the board during discussion
- Give step-by-step directions for labs.
- Prepare word walls or glossary sheets with illustrated vocabulary for students to easily access.
- Summarize discussion and learning more frequently.
- In setting up groups, pair non-native with native speakers.
- Make connections to the students' out of school experiences.
- Vary instructional delivery to include picture books, video, etc.
- Digital Commons
From: Westphal, L.E. (2009). Differentiating instruction with menus: science. Waco, Texas: Prufrock Press, Inc.
- "Food chains control the transfer of energy through an ecosystem. Choose a local food chain and create a PowerPoint presentation to show how the energy is transferred and converted through the system."
- "Research Rube Goldberg and his machines. Create your own Rube Goldberg machine that can move a marble more than one meter, has at least 10 different parts, and has at least three different energy transfers. Present your machine and explain the energy transfers to the class."
Research what materials are used in other cultures to insulate materials. How do people in cold climates keep themselves warm?
"Create a set of trading cards for the different types of energy transfers. Be sure to include a real-world example of each." Westphal, L.E. (2009). Differentiating instruction with menus: science. Waco, Texas: Prufrock Press, Inc.
Parents/Admin
Administrators
- From: SciMathMN Minnesota K-12 Science Framework, 1997:
- Students should work with teacher guidance.
- This standard should be paired with an Inquiry standard whenever appropriate.
- When possible, students should be given opportunities to work in authentic settings.
- Students must demonstrate basic safety procedures and skills when using tools and equipment.
- Administrators should see students drawing or conceptualizing the transfer of energy from one object to another. This may be in the form of a chart, diagram, or graph.
- Watch the video with your kids and take time to experiment with different insulators in your house. Take some boiling water and pour it into a jar that will seal tightly. Wrap it in a down jacket and record the temperature each hour until it reaches room temperature. Do the same with fleece, a cotton sweat shirt, a wool sweater, or any other items that your student may use to keep themselves warm. Look at your data and discuss with your student which materials hold in heat better and why. If a thermometer is not available, the same experiment can be done with an ice cube in a jar and recording how long it takes to fully melt when wrapped in each of the items.
- Take a tour of your house with your student and discuss how heat is kept from dissipating into the outdoors. Examine ice dams on the roofs of your neighborhood during the winter. Research how the dams form and what can be done to prevent them.
